Popular Science Essay | The Hidden Codes in Light’s Colors: How Wavelengths Shape the World’s Hues
![]() 04/07 2026
04/07 2026
![]() 403
403
The Hidden Codes in Light’s Colors: How Wavelengths Shape the World’s Hues
Following a sudden rainstorm, the sky’s azure deepens, and a vibrant rainbow quietly emerges in the distance. As the icy winds of polar night subside, the inky blackness of the sky intensifies, and a faint green aurora gently unfolds at the horizon. After a sandstorm, the boundaries between heaven and earth blur, and the entire world is shrouded in a haze of earthy yellow. 
Rainbow | Image Source: Pixabay
Color Is Not an Inherent Property of Objects
Have you ever considered that the vivid “colors” of our world do not solely depend on the objects themselves? What exactly are the various “colors” we perceive? The “red,” “blue,” and “green” we see are merely a collaborative interplay between light and our eyes, and the underlying logic of this interplay reveals the most romantic secret of optics.
If asked, “What color are tree leaves?” most would instinctively answer, “Green.” Yet, leaves do not possess a fixed “color property.” They contain abundant chlorophyll, whose chloroplasts vigorously absorb red and blue light from sunlight to produce nutrients while reflecting green light. When this reflected green light enters our eyes, our brains interpret it as “the leaves are green.” Transmission also plays a subtle role—for instance, the green hue of leaves stems not only from the reflection of green light wavelengths but also from the transmission of some green light by chlorophyll. This tacit cooperation between reflection and transmission allows us to perceive the vibrant green of leaves.
In essence, the colors we see result from an object “filtering” sunlight. Taking reflection as an example, the color of light an object reflects determines the color we perceive; if an object absorbs all light, it appears black; if it reflects all light, it appears white. In 1666, Isaac Newton unveiled the secret of white light through a simple experiment: by passing a beam of sunlight through a prism, the single beam of white light instantly dispersed in space, forming a colored band of light ranging from red to violet on the wall—the familiar “spectrum.” This experiment demonstrated that white light is not indivisible but composed of various “colored lights.”
Prism Dispersion | Image Source: Pixabay
The “Secret Code” of Colors
Why does light, which is essentially the same, appear in different colors such as red, green, and blue? The answer lies in “wavelength”—each type of light has its own fixed wavelength, akin to a unique ID number. Different wavelengths of light correspond to different colors in our perception. We can envision light as “propagating waves”—light is also an electromagnetic wave, and during its propagation, it exhibits periodic “undulations.” Wavelength is the distance between two adjacent wave crests (or troughs), usually measured in “nanometers” (nm). Within different wavelength ranges, the “visible light” we can see constitutes only a small part of the electromagnetic wave family, with a wavelength range of 380 nm to 760 nm.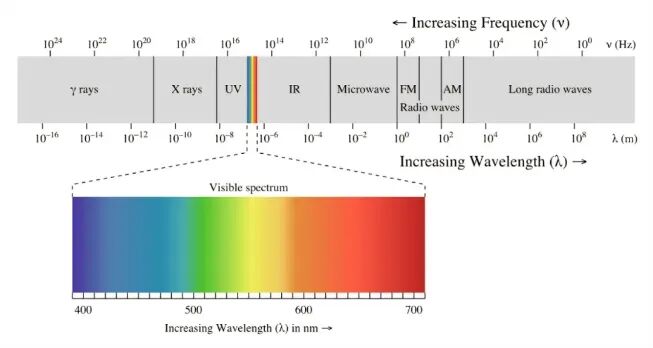
Electromagnetic Spectrum | Image Source
Within the “family” of visible light, each color occupies a clear “exclusive range” of wavelengths. From red to violet, the wavelength of light gradually shortens, forming a complete spectrum.
Red light: With a wavelength range of approximately 620–760 nm, it has the longest wavelength among visible light. Due to its longer wavelength, it is not easily scattered by tiny particles in the air during propagation, granting it strong penetrating power.
Electromagnetic Spectrum | Image Source: Pixabay
Blue light: With a wavelength of about 450–495 nm, it has a shorter wavelength and is easily scattered by dust and water vapor in the air. The blue light emitted by phone and computer screens, due to its short wavelength and relatively high energy, can cause eye strain after prolonged exposure, which is why we need “anti-blue light glasses.” Every color we see is a “signal transmitted” by light of a specific wavelength. For example, the skin of an apple absorbs short-wavelength light such as green and blue light, reflecting only red light, which is why we perceive the apple as red. If the wavelength of the illuminating light is altered, the color of the object will also change: if a red apple is illuminated with pure blue light, since the apple cannot reflect blue light, it will appear black. This further proves that the essence of color is “the result of specific wavelengths of light being reflected,” and without differences in wavelength, there would be no differences in color. Now, let’s explore meteorological phenomena related to color. Why is the sky blue? On a sunny day, the air is filled with tiny molecules such as nitrogen and oxygen (with diameters much smaller than the wavelength of light), and Rayleigh scattering “favors” short-wavelength light—blue light, with its short wavelength, is scattered in large quantities into the sky; red, yellow, and other longer-wavelength lights are scattered weakly and pass directly through the atmosphere, which is why the sky appears blue. At sunrise and sunset, because the sun’s elevation angle is very low, the distance light travels through the atmosphere is longer than at noon. In this case, the shorter-wavelength violet and blue lights weaken more along the way than red, yellow, and orange lights. Therefore, the sky often appears a beautiful reddish-orange color, which is what we see as sunrise and sunset. However, on cloudy days, the sky appears grayish-white and does not display a dominant wavelength color. Why is this? On cloudy days, the clouds are thick, and the air contains a large number of water droplets, ice crystals, and other large particles (with diameters close to or larger than the wavelength of light). Mie scattering has little difference in scattering various wavelengths of light, and all colors of light are scattered uniformly. When multiple colors of light mix, the sky appears grayish-white. The light we commonly encounter in life is actually electromagnetic waves, differing only in wavelength. Wavelength differences determine their respective uses and forms of presentation. Electromagnetic wave bands beyond visible light are also crucial: on the short-wave end, from short to long, are hard X-rays and soft X-rays. The former is used for industrial inspection and severe medical diagnosis due to its strong penetrating power, while the latter is suitable for biomedical imaging; extreme ultraviolet light is the core light source for chip lithography, while deep ultraviolet and near-ultraviolet lights play roles in sterilization, lithography development, and fluorescence detection, respectively. Near-infrared light with wavelengths of 760–1400 nm is commonly used in TV remote controls to emit infrared signals for device control, while longer-wavelength far-infrared light is integrated into heaters and wellness devices. Microwaves (with wavelengths of 1 mm to 1 m), which have even longer wavelengths than infrared light, can be used to heat food in microwave ovens and to transmit signals for mobile phone communications. Terahertz waves (with wavelengths of approximately 1000–100 μm), which lie between microwaves and infrared light, are often received using superconducting detectors or photonic crystal antennas, enabling precise capture of weak signals and providing critical data for material imaging, security detection, and other applications.
The Colors of Life—The Color Principles of Displays
Since the essence of color is “the result of specific wavelengths of light being reflected,” how do the digital products surrounding us produce color? Displays can present a myriad of colors based on the “principle of the three primary colors of light”—the mixing of light is an “additive mixture,” and red (R), green (G), and blue (B) wavelengths of light can be combined to form the “basic units” of almost all colors perceivable by human vision. The key to the principle of the three primary colors of light is that the cone cells in the human retina are most sensitive to these three wavelengths of light. When these three types of light are superimposed in different intensities, the brain “translates” them into different colors—for example, red light + blue light = purple light.
Displays utilize this principle by precisely controlling the intensity of RGB light to “deceive” our visual system and present a rich array of colors. The display images we see are composed of countless “pixels,” with each pixel acting as a “miniature RGB color palette.”
Pixel Diagram | Image Source: Pixabay
In an LCD display, each pixel contains three closely adjacent “sub-pixels,” corresponding to red, green, and blue filters. Behind the screen is a “backlight layer.” When white light passes through different sub-pixels, liquid crystal molecules act like “valves,” controlling the intensity of light passing through the filters. To display red, only the white light is allowed to pass through the red filter, while the valves for the green and blue sub-pixels are closed. The different intensity ratios of the three sub-pixels result in different colors for the pixel as a whole, and the combination of countless pixels forms the complete colored image we see. Compared to LCD displays, OLED displays do not require a backlight layer; each sub-pixel can emit light on its own—when the red sub-pixel is powered on, it directly emits red light. By adjusting the current flowing through each sub-pixel, the brightness can be controlled, thereby mixing to produce the desired color. In this structure, the pixel’s color response is faster, and “pixel-level light control” can be achieved. We may wonder how gradient colors, such as light red to deep red, cannot be simply achieved with sub-pixels in only two states (“on”/“off”). Therefore, we also need “gray levels”—that is, the “brightness levels” that sub-pixels can display. For example, a common “8-bit gray level” means that each sub-pixel has 28 = 256 brightness levels (ranging from darkest to brightest in 256 steps). Thus, the number of colors a single pixel (RGB three sub-pixels) can display is 256 × 256 × 256 ≈ 16.77 million, which is sufficient to meet the color needs of daily office work and entertainment.
The Colors of Scientific Research—Lasers
The color of light is uniquely determined by its wavelength. Ordinary light sources appear multicolored because they contain light of multiple wavelengths. Are there any light sources with particularly good monochromaticity? The answer is yes—lasers. One of the core characteristics of lasers is their monochromaticity—the light they emit has an extremely concentrated wavelength, containing almost only light of a single “pure wavelength.” This extreme “wavelength concentration” gives lasers an exceptionally pure color.
Laser Show | Image Source: Pixabay
The degree of monochromaticity is also gauged by the concept of 'linewidth.' A narrower linewidth signifies a more concentrated wavelength range of light, and thus, a purer color. The linewidth of conventional light sources is quite broad. For instance, the visible light wavelength range of sunlight spans approximately 380-760 nm, resulting in a linewidth of 380 nm. This means sunlight effectively 'emits all colors of light simultaneously.' Even LED lights, which appear to have very pure colors (such as red LEDs), possess a linewidth of 20-50 nm, and are mixed with a trace of orange or deep red light. In stark contrast to ordinary light sources, lasers boast an extremely narrow linewidth. A typical helium-neon laser, for example, has a linewidth of only 0.00001 nm, practically 'emitting only red light at 632.8 nm.' For precision experiments, frequency-stabilized lasers can achieve a linewidth as narrow as 10-12 nm. How can lasers attain such a narrow linewidth? The secret lies in the principle of laser generation—stimulated emission. During the laser emission process, atoms are guided by an 'external optical signal' to emit photons in unison. These photons possess completely consistent wavelengths and phases, akin to 'soldiers marching in formation.' In contrast, the emission of ordinary light sources is 'spontaneous emission,' where atoms randomly emit photons with jumbled wavelengths, naturally leading to impure colors.
The monochromaticity of lasers not only provides a 'precise probe' for scientific research to delve into the microscopic world but also fulfills the core requirements of various practical applications. In scientific research, ultrafast spectroscopy utilizes monochromatic laser light to supply specific energy photons, precisely exciting specific electronic states or vibrational modes in materials. This aids in the study of ultrafast processes such as superconducting electron pairing and semiconductor carrier relaxation. Angle-resolved photoelectron spectroscopy relies on the monochromaticity of lasers to enhance energy resolution, clearly measuring the electronic band structures of topological insulators and high-temperature superconductors, and revealing their unique quantum properties. Raman spectroscopy also benefits from single-wavelength laser light, which minimizes background interference and enables precise analysis of material lattice vibrations and molecular bond vibrations, delving deep into microscopic structures. Lasers have emerged as a pivotal force in breaking through technological bottlenecks and exploring the microscopic world of matter.
In other fields, in optical communications, monochromatic laser light at 1550 nm, due to its concentrated wavelength, mitigates 'wavelength dispersion' caused by variations in the transmission speeds of different wavelengths of light. This enables uninterrupted transmission over thousands of kilometers and supports global internet data exchange. In the medical field, 532 nm green laser light is selectively absorbed by melanin, allowing for precise freckle removal without damaging the surrounding skin. A 193 nm excimer laser can act like a 'precision carving knife,' emitting laser light solely in the preset corneal area (Note: 'preset' here refers to a predetermined or programmed area). It precisely breaks the molecular bonds of corneal tissue, vaporizing the parts that need to be removed without affecting the surrounding non-preset corneal area. This ensures that the shape and depth of the incision fully meet the requirements for correcting myopia and achieving surgical precision.
In industrial processing, 193 nm deep ultraviolet monochromatic lasers can precisely etch nanoscale circuits on semiconductor chips. Meanwhile, 1064 nm infrared monochromatic lasers can be focused into micrometer-scale spots for precise drilling and welding of metals without damaging other areas of the material.

Laser Show | Image Source: pixabay
Electromagnetic waves, with wavelength as their fundamental metric, have established a comprehensive application system that permeates all aspects of life. The visible light spectrum achieves color presentation through wavelength combinations, supporting display technologies. Meanwhile, the invisible electromagnetic waves at both ends of the spectrum expand their functionality with unique physical properties. It is the variations in wavelength that endow various electromagnetic waves with distinct functional attributes, ultimately transforming intangible electromagnetic radiation into the core technological foundation that underpins modern life. Light serves as both a precise key to decoding the laws of the universe and an invisible brush that weaves romance into life. It allows the sky to reflect a clear blue, the rainbow to bloom with seven colors, and the screen to flow with countless light and shadow. Between rational exploration and emotional aesthetics, it becomes a unique link connecting science and poetry.






