Ultra-fast Nonlinear Optical Technology: A 1 GHz Mid-Infrared Frequency Comb for Rapid Spectral Analysis
![]() 03/05 2025
03/05 2025
![]() 933
933
The key to gaining deeper insights into chemical reaction pathways lies in having a controllable setup capable of in-situ detection. An ideal in-situ detection method must detect numerous chemical species with high specificity, accuracy, and operate within an appropriate reaction time scale. This article reports on a 1 GHz dual-comb spectroscopy system operating in the mid-infrared range that meets these requirements [1].
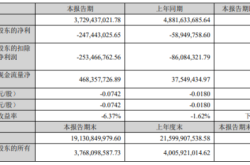
Figure 1 compares three primary mid-infrared frequency comb architectures. Quantum cascade/interband cascade (QCL/ICL) frequency combs have tooth spacings greater than 10 GHz, offering high acquisition rates and comb tooth power but limited single-shot resolution and spectral bandwidth. Electro-optic modulation frequency combs have narrower tooth spacings (e.g., 300 MHz), enabling higher resolution but with limited spectral bandwidth. Mode-locked laser frequency combs typically maintain high pulse peak powers for driving nonlinear optical processes, achieving broader spectral bandwidths but with slightly lower repetition rates. Figure 1(e) compares the performance of existing mid-infrared dual-comb spectrometers used in gas-phase spectroscopy experiments.
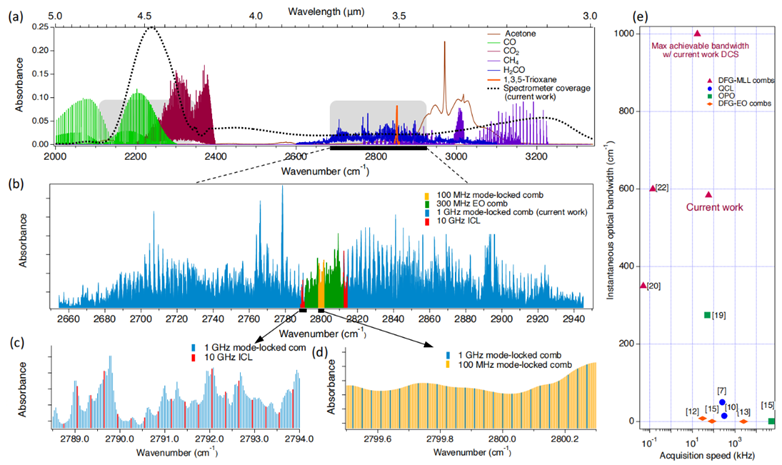
Figure 1 Comparison of Various Mid-Infrared Frequency Comb Spectrometers [1]
The work presented in this article builds upon previous research by the same group. Figure 2 shows the experimental setup completed in 2022 [2]: A 1 GHz mode-locked laser with a center wavelength of 1.56 μm serves as the seed source. The setup includes two stages of all-fiber amplifiers (a core-pumped EDFA and a clad-pumped EYDFA), a grating pair to compress the pulses, and a segment of highly nonlinear anomalous dispersion fiber for spectral broadening. Mid-infrared optical pulses are generated via intra-pulse difference frequency generation in a nonlinear crystal. The spectrum's evolution throughout the process is shown below Figure 2. Using a PPLN crystal, a 1 GHz mid-infrared frequency comb with a wavelength range of 3-5 μm and an average power greater than 4 mW can be produced.
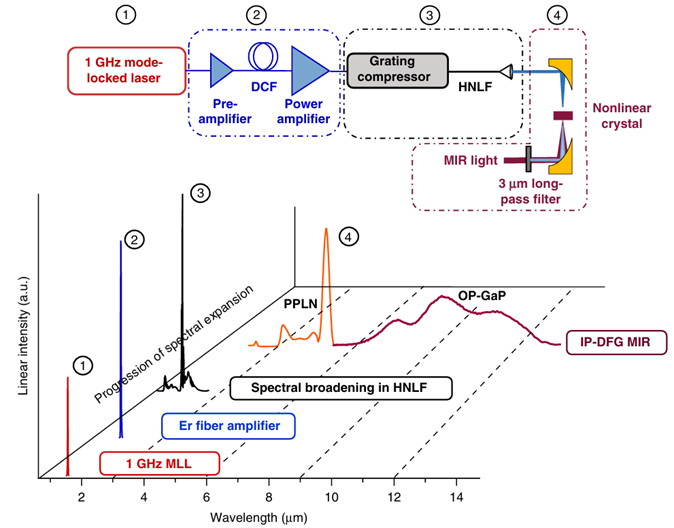
Figure 2 Generation of a Broadband 1 GHz Mid-Infrared Frequency Comb Source via Intra-Pulse Difference Frequency Generation [2]
The research group constructed a dual-comb spectroscopy system using two optical frequency combs, as shown in Figure 2, to study the decomposition of 1,3,5-trioxane into formaldehyde and the subsequent formation of carbon monoxide (CO) from formaldehyde. After passing through the shock tube, the dual-comb light source is split into two parts by wavelength, with each part entering its respective detector, as illustrated in Figure 3. This measurement method expands the spectral bandwidth at a given acquisition rate, enhancing the signal-to-noise ratio. Using an FPGA data acquisition card, interferograms are sampled at a 1 GHz rate, capturing one interferogram every 17.5 μs.
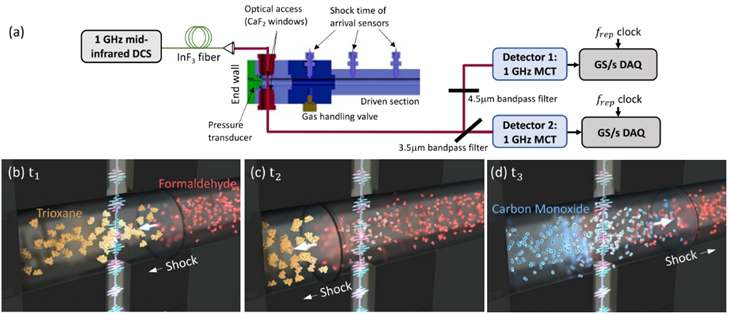
Figure 3 Experimental Setup [1]
Figure 4 (top) shows the mid-infrared light source outputting a spectral width of 1800 nm, covering the absorption bands of trioxane, formaldehyde, and carbon monoxide. Researchers recorded the transmission spectrum of trioxane before and after the chemical reaction shockwave reached the reflection surface, with a time step of 17.5 µs. The concentration of chemical species and reaction temperature at a specific moment are akin to a "frame" in a movie. Connecting multiple such "frames" resembles a "molecular movie" of the chemical reaction process. Figure 4(a) shows the average absorption spectrum and data fitting results of trioxane collected before the reflection of the reaction shockwave. Figures 4(b) and (c) present the average absorption spectra and data fitting results of formaldehyde (t=17.5 µs) and CO (t=52.5 µs) after the reflection.
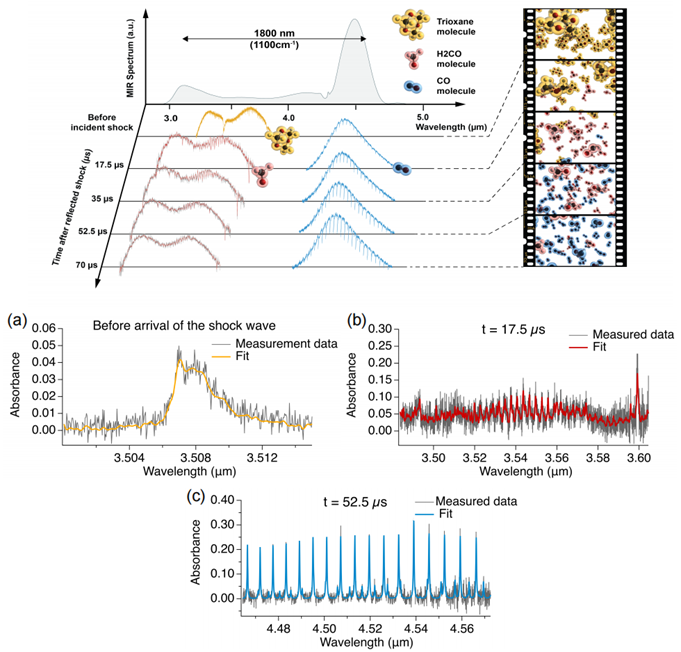
Figure 4 Detection of the Decomposition Reaction of 1,3,5-Trioxane Using Dual-Comb Spectroscopy [1]
Figure 5 displays the measured temperature, concentration, and pressure results from a set of experiments. The black solid lines represent simulation results based on the created chemical kinetic model. The CO concentration reaches a maximum and stabilizes before the temperature begins to decline. The measured temperature aligns with the simulated temperature curve within the uncertainty range, and the simulated and measured CO concentrations are in good agreement.
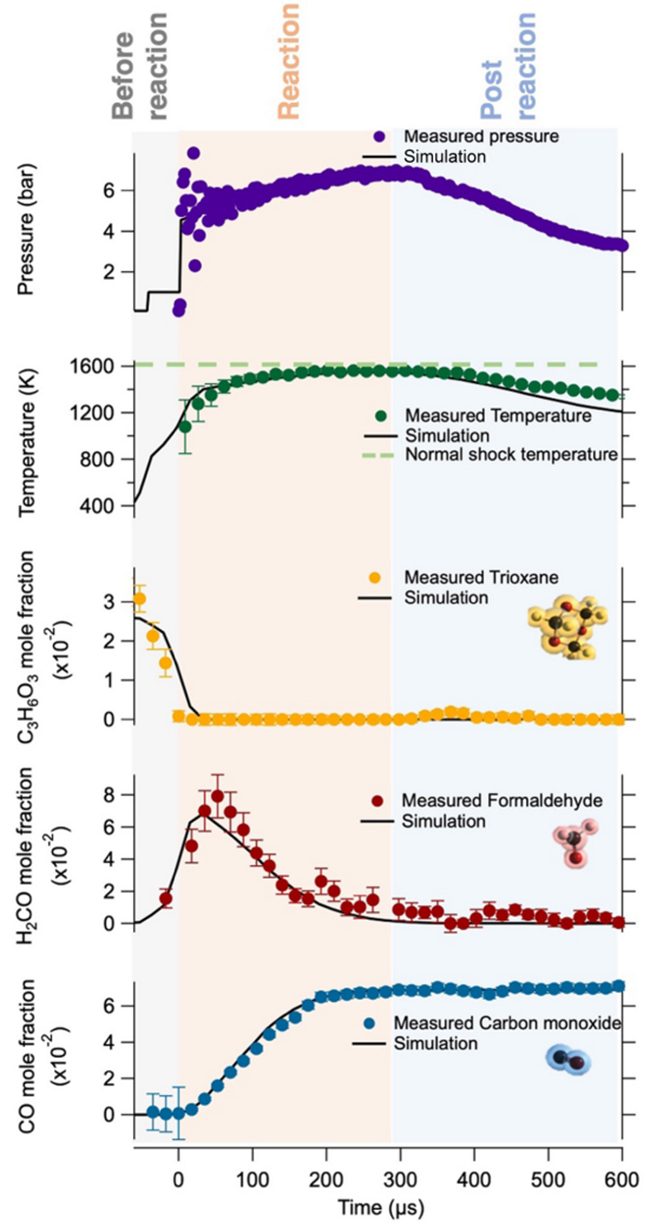
Figure 5 Reaction Progression: Changes in Temperature, Pressure, and Chemical Species Concentrations Over Time [1]
In summary, researchers have employed a 1 GHz mid-infrared dual-comb spectroscopy system for the first time to study the decomposition process of trioxane, achieving high bandwidth (3-5 μm, >1000 cm⁻¹), high spectral resolution, and high acquisition rates, which holds significant application prospects.
References:
[1] N. Hoghooghi, P. Chang, S. Egbert, M. Burch, R. Shaik, S. A. Diddams, P. Lynch, and G. B. Rieker, "GHz Repetition Rate Mid-Infrared Frequency Comb Spectroscopy of Fast Chemical Reactions," Optica 11(6), 876 (2024).
[2] N. Hoghooghi, S. Xing, P. Chang, D. Lesko, A. Lind, G. Rieker, and S. Diddams, "Broadband 1-GHz Mid-Infrared Frequency Comb," Light Sci Appl 11(1), 264 (2022).