Active Tracking AOOCT for Single-Cell Retinal Functional Imaging in Medical Optical Technology
![]() 05/07 2026
05/07 2026
![]() 410
410
In the realms of ophthalmic imaging and visual neuroscience, scientists have long pursued the goal of non-invasively observing neural activity in the living retina with high resolution. Traditional methods for detecting neural activity with high sensitivity, such as fluorescent probes and microelectrode arrays, face limitations in clinical applications due to their invasive nature.
In recent times, optical retinography (Optoretinography, ORG) has emerged as a promising non-invasive optical measurement technique. When cone cells are stimulated by light, they initially undergo rapid contraction followed by slow elongation, leading to nanoscale changes in the optical path length. By leveraging adaptive optics optical coherence tomography (AOOCT), researchers can harness the phase information of light to capture, with nanoscale sensitivity, the minute physical deformations in the outer segments of photoreceptor cells in response to light stimulation.
A significant challenge in achieving in vivo single-cell functional imaging in clinical settings is the uncontrolled movement of the eye. To extract extremely weak signals using optical coherence tomography (OCT), the probe must maintain extreme stability on the same cell. However, even during steady fixation, the eye inevitably drifts and experiences microsaccades. Traditional strategies involve capturing large-field images through ultra-high-speed three-dimensional volume scans and compensating for eye movements through time-consuming and complex offline image spatial registration. The drawbacks of this approach include high computational costs, limited temporal resolution, and a compromised signal-to-noise ratio.
This paper presents a breakthrough in retinal ORG imaging by introducing a hardware-based active eye-tracking stabilization technology. This technology eliminates the need for offline data processing and enables real-time, active guidance of the OCT beam during imaging [1]. To achieve active stabilization, the research team designed a system that integrates adaptive optics scanning laser ophthalmoscopy (AOSLO) and adaptive optics optical coherence tomography (AOOCT). The system also incorporates adaptive optics technology to compensate for higher-order aberrations in the human eye, significantly enhancing retinal imaging resolution. A schematic representation of the system setup is depicted in Figure 1. 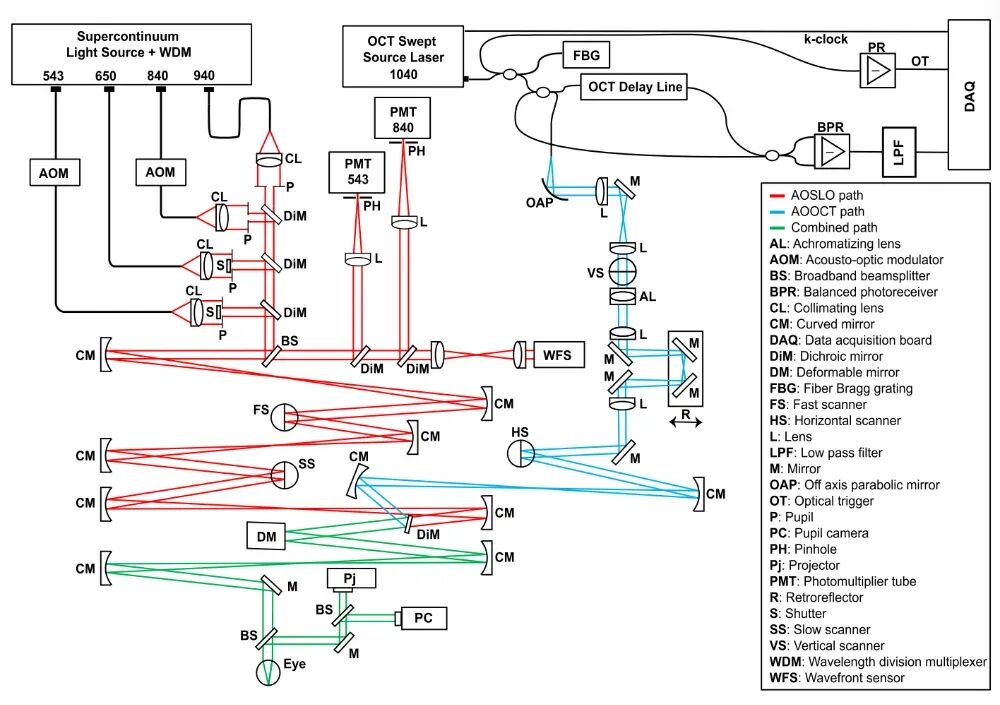 Figure 1. Active Stabilization ORG Imaging System Based on AOSLO-AOOCT [1]
Figure 1. Active Stabilization ORG Imaging System Based on AOSLO-AOOCT [1]
AOSLO facilitates high-precision eye-tracking functionality, acquiring high-resolution retinal images in real-time at a frame rate of 30 Hz. The system divides the real-time scanned images into 32 stripes and performs high-speed cross-correlation registration calculations stripe by stripe with a pre-acquired reference frame. This allows for continuous tracking of the eyeball position at a frequency of up to 960 Hz (30 frames × 32 stripes), as illustrated in Figure 2. The calculated eye movement offsets are directly converted into tracking signals and transmitted in real-time to a high-speed scanning mirror in the AOOCT beam path to compensate for eye movements, thereby anchoring the beam to specified retinal patches or even single cells. 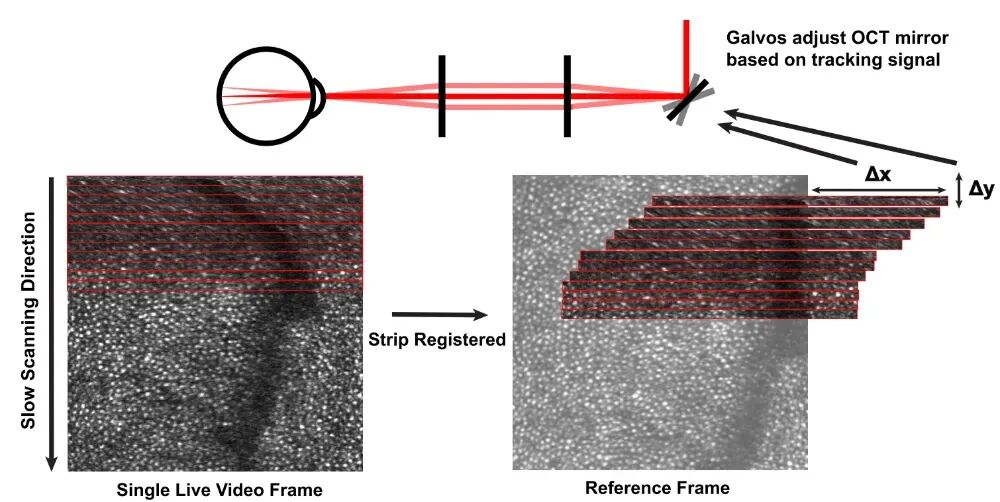 Figure 2. Schematic of AOSLO Eye Tracking [1]
Figure 2. Schematic of AOSLO Eye Tracking [1]
Thanks to precise eye movement compensation, the system offers versatile ORG scanning modes, as shown in Figure 3. Mode A involves point scanning, where the probe locks onto a single cone cell for up to 3.57 seconds, unleashing the ultimate temporal resolution of OCT at 100 kHz. Mode B entails line scanning, which repeatedly scans an extremely short line segment, striking a balance between high temporal resolution and high signal-to-noise ratio achieved through spatial averaging. Mode C involves volume scanning, which performs targeted three-dimensional mapping of small clusters of cells.  Figure 3. Three Scanning Modes of the ORG System [1]
Figure 3. Three Scanning Modes of the ORG System [1]
Utilizing this active stabilization system, this paper conducted ORG imaging on the retina of a subject. In both Mode A and Mode B, the system successfully captured the rapid contraction and slow elongation phase responses of cone cells following stimulation with 543 nm green light—observations that were previously extremely difficult to make clearly due to eye movement blur in older systems. 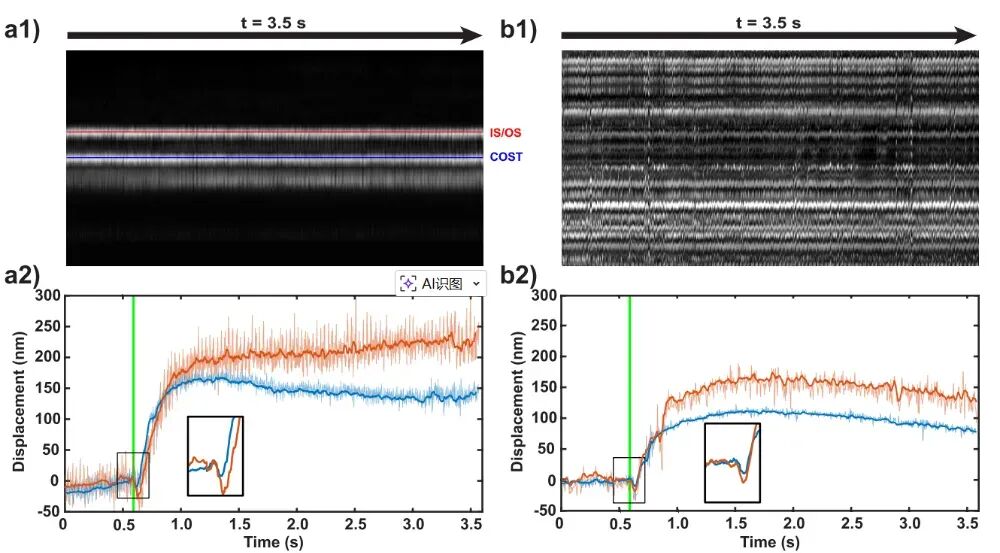 Figure 4. Length Changes in Cone Cells Following Light Stimulation Measured by Mode A (Left) and Mode B (Right) Scanning [1]
Figure 4. Length Changes in Cone Cells Following Light Stimulation Measured by Mode A (Left) and Mode B (Right) Scanning [1]
The system also achieved high-precision classification of cone cell subtypes. Different types of cone cells (L, M, and S types) exhibit distinct responses to specific wavelengths of light. Using ORG signals acquired in Mode C, this paper classified the subject's cone cell population and compared it with a previously obtained cell classification map derived through complex traditional methods, achieving a remarkable match rate of 90.1%. 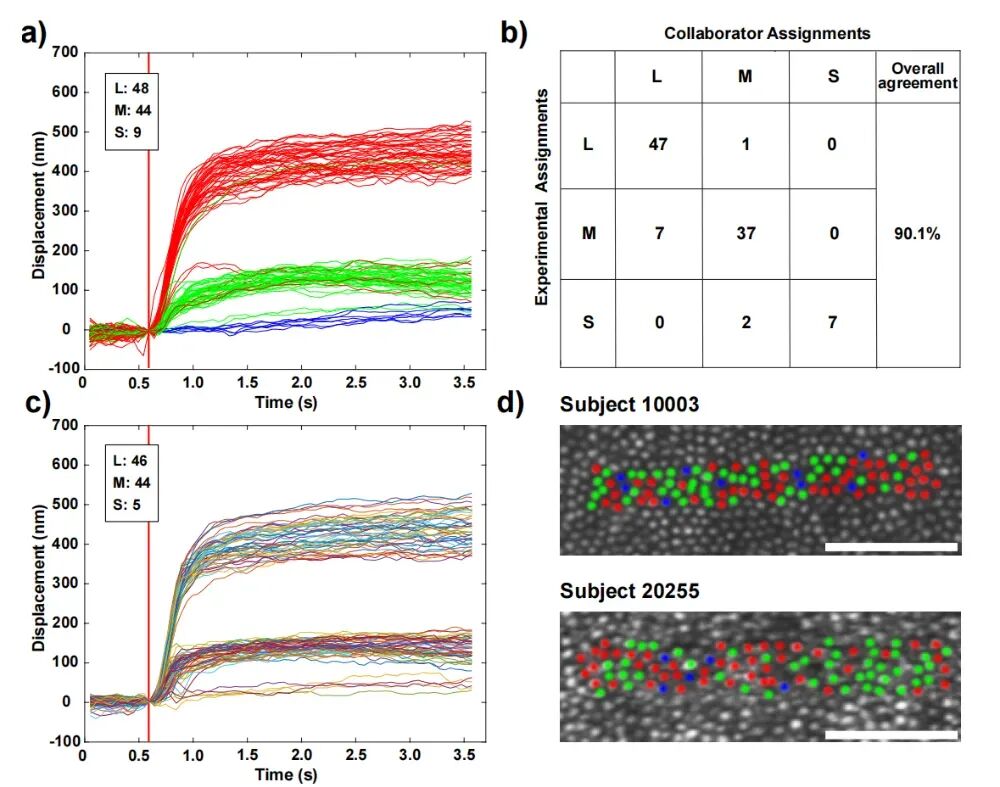 Figure 5. Classification of Cone Cells by Mode C ORG Scanning [1]
Figure 5. Classification of Cone Cells by Mode C ORG Scanning [1]
Actively stabilized AOOCT provides an unprecedented tool for deepening our understanding of retinal function. The research team states that future objectives will include the challenging task of detecting retinal ganglion cell action potentials with even smaller deformation scales (at the single-nanometer level) and extremely rapid responses (at the millisecond level), ultimately enabling non-invasive mapping of the extremely fine neural networks processing visual information in the living human eye.
References: [1] Wong, J. H., et al., Optoretinography with actively stabilized adaptive optics optical coherence tomography. Biomed Opt Express. 16(8): 3222-3236 (2025).